Computer Systems Validation, Part 2: Implementation
Course Summary
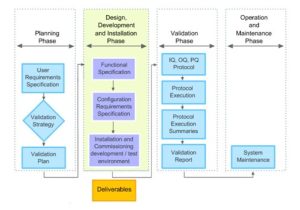
This module describes the design, development and installation phase, the validation phase, and the operation and maintenance phase of the validation of computerised systems in medicines and healthcare products manufacturing environments. It continues to follow the progress of a pharmaceutical company’s project to validate a new dispensary control system.
£99.00 Original price was: £99.00.£69.00Current price is: £69.00. exc. VAT
Purchasing Information
When you have completed your course order, Zenosis will finalise the setup of your course materials and contact you on the email address that you provide in your order. You can expect this process to be completed within one business day (using the UK business calendar) of completing your payment.
It is therefore essential that you use your real email address for your order, or indicate in the purchase notes the email address to be used for the course set-up, and check that any messages from Zenosis or grapl are not lost in your junk or spam folder.
You will have access to the course module(s) for a period of 180 days after your purchase is complete.
Detailed Course Information
• Describe the design, development and installation phase of projects to validate computerised systems
• Describe the validation phase of such projects
• Describe the operation and maintenance phase
• Determine which systems to validate
• Determine the amount of validation required, and the strategy to use
Manufacturing personnel in the pharma/biotech, dietary supplement, and medical devices industries need to understand the principles and practice of computer systems validation, as set out in this module. In particular, the module provides essential learning for engineering, information and communication technology, production, and quality management personnel in the pharmaceutical industry.
• Design, development and installation phase
– This session specifies the roles of functional and design specifications. It outlines the development testing process, and describes the formulation and use of test plans, cases and scripts. It identifies characteristics of good testing practices, and emphasises the importance of development change management.
• Validation phase
– This session specifies the activities to be performed in the validation phase, and outlines their timing. It states the purposes of platform qualification, application installation qualification, operational qualification, and performance qualification. It specifies tests typically carried out in operational qualification and performance qualification. Finally, it describes the roles of validation change management and the validation report.
• Operation and maintenance phase
– This session describes the measures that need to be in place during the operation and maintenance phase. It outlines the management of the decommissioning of a system. It identifies changes that need to be controlled in the operation and maintenance phase.
• Assessment
– The assessment tests the learner’s assimilation of the module’s content.
